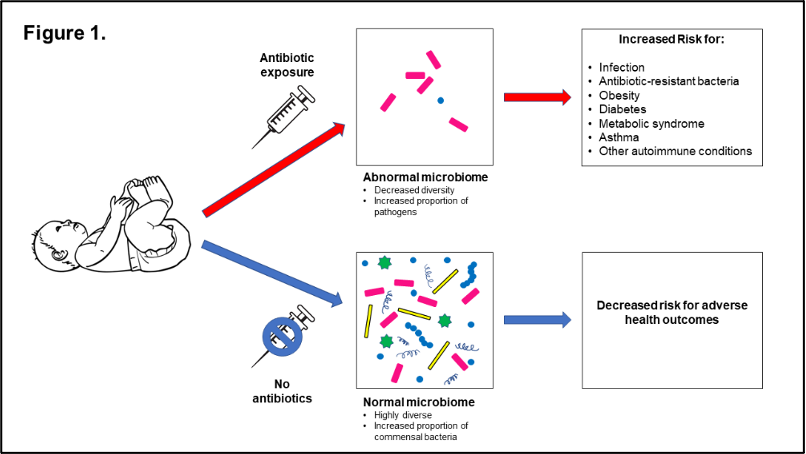
Antibiotic exposure in neonates is associated with significant morbidity and mortality. Infants who receive antibiotics, particularly within the first few weeks of life, are at increased risk for a variety of short- and long-term adverse outcomes.
These adverse effects are mediated by disruption of the normal infant microbiome following antibiotic administration (Figure 1). Following a course of antibiotics, the microbiologic diversity of the gut and lung microbiome is reduced and pathogenic organisms such as Escherichia coli become the predominant organisms. This loss of diversity and “pathogen surge” leads to increases in inflammation and risk for infection in the short term and increased energy harvest and weight gain in the long term. As a result, antibiotic exposure in newborns has been associated with increased risk for infection from antibiotic-resistant organisms, asthma, obesity, and metabolic syndrome in later childhood.
Similarly, the alteration of the normal gut and lung microbiome has implications for immune development. Normal gut and lung flora have an innate anti-inflammatory effect; loss of this inflammatory suppression leads to increased airway reactivity (e.g., increased risk for asthma or infection-associated wheezing) and decreased immune tolerance (e.g., increased risk for eczema, type 1 diabetes, celiac disease, and other atopic or autoimmune conditions). These adverse outcomes can be seen after even a single dose of antibiotics.